FDA ESG NextGen: What It Means for Medical Device and SaMD Companies
- luminawebsitedesig
- Apr 23
- 3 min read
By Shannon Campbell, PhD, Principal Consultant, Frank Healthcare Advisors
The U.S. Food and Drug Administration has taken a significant step toward modernizing regulatory infrastructure with the rollout of the Electronic Submissions Gateway Next Generation (ESG NextGen). While at first glance this may appear to be a technical upgrade, it represents a broader shift toward digitally enabled, scalable, and automation-ready regulatory interactions; a shift that is particularly relevant for companies developing software as a medical device (SaMD) and AI/ML-enabled technologies.
For small and mid-sized innovators, understanding how ESG NextGen fits into the broader FDA submission ecosystem, and how to operationalize it efficiently, will be increasingly important.
Key Takeaways:
ESG NextGen is the FDA’s modern, cloud-based submission gateway
It enables higher capacity, automation, and improved security
It complements, rather than replaces, eSTAR and the CDRH Portal
It is particularly relevant for data-intensive and AI-enabled devices
Companies should begin planning for digitally scalable regulatory workflows
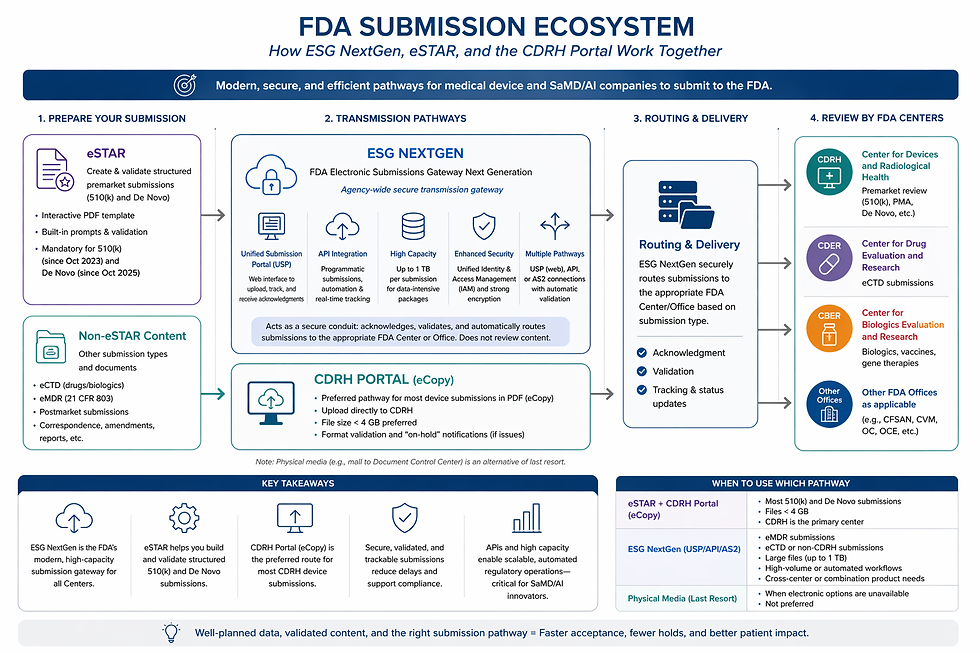
What is ESG NextGen?
ESG NextGen is the FDA’s modernized, cloud-based platform for receiving and routing electronic regulatory submissions. It replaces legacy systems (such as WebTrader) with a more scalable, secure, and flexible infrastructure.
At its core, ESG NextGen functions as a secure transmission gateway by:
Receiving electronic files from sponsors
Performing validation and acknowledgment
Routing submissions to the appropriate FDA Center (e.g., CDRH, CDER, CBER)
ESG NextGen serves as a “unified entry point for all submission types,” with significantly enhanced capacity and improved performance for high-volume or data-intensive workflows.
Key Features and Capabilities
1. Unified Submission Portal (USP)
A web-based interface replaces the legacy WebTrader system, allowing users to upload submissions, track submission status, and receive acknowledgments across FDA Centers. This improves usability, particularly for organizations without dedicated regulatory IT infrastructure.
2. Multiple Submission Pathways, Including API-Enabled Submissions
One of the most important upgrades is API integration, which enables:
Automated submission workflows
Integration with internal regulatory systems
Real-time status tracking
Companies can also submit via Web portal (USP) or AS2 connections.
Each pathway includes automated validation and acknowledgment workflows, reducing submission uncertainty and delays. For AI/ML companies operating continuous development cycles, this opens the door to more scalable regulatory operations.
3. High-Capacity Data Handling
ESG NextGen supports submissions up to 1 terabyte, a major increase over legacy constraints. This is particularly relevant for:
Imaging-heavy SaMD (e.g., radiology, cardiology platforms)
AI/ML training or validation datasets
Real-world evidence (RWE) packages
4. Enhanced Security and Identity Management
The platform introduces a unified Identity and Access Management (IAM) system, simplifying user credential management, organizational access control, and security compliance
ESG NextGen in Context: Not a Replacement for eSTAR or CDRH Portal
A critical point for medical device companies is that ESG NextGen does not replace existing CDRH-specific tools, but rather complements them.
Tool | Role |
eSTAR | Content creation and validation (mandatory for 510(k) and De Novo) |
CDRH Portal | Preferred upload pathway for most device submissions |
ESG NextGen | Agency-wide transmission gateway |
For most SaMD companies:
eSTAR + CDRH Portal will remain the primary pathway for premarket submissions
ESG NextGen becomes relevant for:
Large or complex submissions
Cross-center interactions (e.g., combination products, CBER involvement)
Automated or high-frequency submission environments
Practical Considerations for Companies
For small and mid-sized device developers, the key question is not whether to use ESG NextGen, but when and how to incorporate it into your regulatory strategy.
Recommended approach:
Continue using eSTAR + CDRH Portal for standard device submissions
Evaluate ESG NextGen if you:
Manage large datasets or imaging-heavy submissions
Plan to scale submission frequency
Want to automate regulatory workflows
Interact across multiple FDA Centers
FHA Perspective
At Frank Healthcare Advisors, we view ESG NextGen as more than a technical upgrade, it is part of a broader FDA transition toward digitally integrated regulatory ecosystems.
For innovators in SaMD and AI/ML:
Regulatory strategy must now consider not just what you submit, but how you submit it
Early alignment of data structure, validation outputs, and submission pathways can reduce downstream friction
Scalable regulatory infrastructure is becoming a competitive advantage, not just an operational detail



Comments